Asbestos lurking in baby talc powder??
For decades, talcum powder companies marketed their products as safe. But is it?
Johnson & Johnson will no longer sell its talc-based baby powder globally in 2023, more than two years after it stopped selling the product in the United States and Canada. The decision to fully stop selling the product came after the FDA carried out tests that found asbestos in a single lot of J&J baby powder, despite the company’s continued statements that its baby powder is safe.
The company has also been ordered to pay billions of dollars with more than 65,000 active lawsuits in verdicts to women and their families who say asbestos-contaminated baby powder caused them to develop ovarian cancer and mesothelioma.
So how did they get away with this for so long and why wasn’t it recalled earlier?
Reuters asserts that the FDA has allowed the talc industry to set their own safety rules for their products over the last 50 years.
“This is the issue with big corporate companies in the pharmaceutical industry. Consumers put their trust in big pharmaceutical companies however the core of the company is money at the expense of ones health”

Facts about marine and ocean pollution | Source: EcoWatch
Reuters uncovered many controversial FDA actions when digging through Johnsons and Johnsons history.
Here is a timeline of important FDA decisions:
- 1973: The FDA finds asbestos in a sample of J&J’s Shower to Shower powder, but never announces the finding.
- 1973: J&J, along with other talc companies and the Cosmetic Toiletry and Fragrance Association (CTFA), convinces the FDA that they could monitor the safety of their own products. The FDA ends plans to test talc samples and the CTFA publishes its own test for the companies to use.
- 1986: A graduate student who learned talc deposits are often laced with asbestos petitions the FDA to require a warning on talc powders. Using information from J&J, the FDA determines this warning is not needed.
- 1994: The FDA receives a request from Dr. Samuel Epstein, a University of Illinois environmental medicine professor, to put a warning label on talc powders. Epstein’s research showed talc itself could lead to ovarian cancer. The FDA denies the petition.
- 2006: The World Health Organisation’s International Agency for Research on Cancer said using talc in the pelvic region was “possibly carcinogenic.” Dr. Epstein files a second petition to the FDA. Despite protests from J&J, the FDA finally performs its own tests. While no asbestos was found, the lab that tested the sample had no prior experience testing for asbestos in talc
- 2011: US authorities fined Johnson & Johnson $70 million for bribing doctors in Europe and paying kickbacks for contracts

2013: a statement from Johnson & Johnson implicitly recognises the possibility that the company’s talc could have asbestos in earlier times.
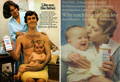
For decades, Johnson & Johnson has promoted its iconic Baby Powder as a safe, gentle product for babies and adults alike.
Known link between talc and ovarian cancer
Talc is considered the softest mineral in the world, it is used across the cosmetic/hygiene industry in everyday products due to its highly stable nature, lack of odour, and the way it absorbs into the skin.
As of now, the main concerns over talc and cancer are the mineral’s associations with ovarian and lung cancer. Women and babies that use the talc may be at an increased risk for ovarian cancer from using J&J talc
According to Globoccan, 295,414 cases of ovarian cancer have been identified in 2018 with a 5 year survival rate of roughly 47%.
Questions were raised with possible link to ovarian cancer dating back as far as the 1960’s, according to the FDA. Instead of warning its consumers, Johnson & Johnson doubled down, including marketing in the 1990’s that targeted minority communities such as African American and Hispanic women. Johnson & Johnson initially started marketing its “best for you”, particularly mothers and babies. Over the years it grew a strong base of branding through its heavily marketed baby products, by building trust with the consumers, despite knowing as early as 1971 that the branding could be tainted with the extremely dangerous human carcinogen asbestos.
One of the very first Johnson & Johnson ads featuring a mother and child was in ‘Wonderful mother ad’ from 1922, still one of their most popular ads till this very day. An ad inspired by a quote from Abraham Lincoln, celebrating the special role that mothers play in their children lives.
Other baby powder adverts used the same strategy, featuring slogans such as ‘a luxury for your baby’ and ‘best for baby - best for you’
Johnson & Johnson's response
Johnson and Johnson denies that the talc powder causes cancer.
In September 1997 internal letter medical consultant wrote suggests that Johnson & Johnson was well aware of the danger its products allegedly posed but chose to do nothing. If the risks are denied, the letter stated,"... the talc industry will be perceived by the public like it perceives the cigarette industry: denying the obvious in the face of all evidence to the contrary.”
Johnson & Johnson released the following statement in an official press release:
“Johnson & Johnson remains steadfastly confident in the safety of talc-based Johnson’s Baby Powder. Decades of scientific studies by medical experts around the world support the safety of our product.”
The company also noted that it will defend the product’s safety in future court cases.
If you think your long-time use of a talc-based product may have contributed to your cancer diagnosis, it would be beneficial to consult with a products liability attorney who would seek justice on your behalf
Sources
- https://www.motleyrice.com/blogpost/put-jj-baby-powder-corner
- https://www.talcumpowdercancerguide.com/
- https://www.reuters.com/investigates/special-report/
- https://ww.fashionnetwork.com/news/johnson-johnson-fined-for-bribing-doctors,165356.html
Conclusion
The one and only way to truly solve this issue is to dramatically reduce the production of plastic, which means curbing our lifestyle from it. 'The most important thing we must do is stop plastic from getting into the ocean in the first place, because it is not feasible or cost-effective to do large-scale cleanups,' says Lau from China Dialog Ocean. “Once in the ocean, plastic waste will stay there for hundreds of years or longer. That is not a legacy I would want to leave for future generations.”
Join the community
Each one of us can make a difference. Together we can make a change. Follow us and subscribe to our Magical Wool Instagram channel as well as our Facebook Page and discover all the latest promotions, news, updates and more.
Popular Stories
SIGN UP
Join our ever growing family who are receiving our latest news, promotions and offers.
COPYRIGHT © 2023 MAGICAL WOOL



